In this article, we summarise the most interesting presentations from the 2026 Berlin conference on myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Genetic evidence pointed towards neural tissue, whole-body scans highlighted muscles and bone marrow, while trials on immunoadsorption, low-dose naltrexone, and methylprednisolone reported null results.
Full videos of the presentations will be made available on the website of the ME/CFS Research Foundation. Big thanks to them for organizing this important event!

Introduction
This was already the fourth scientific conference on ME/CFS in Berlin, and it’s growing every year. This edition, there were 52 speakers and chairs from 10 countries, 54 scientific posters, and more than 6000 online participants. The conference is organized by the Charité fatigue center in Berlin in a historically important building for science: the Harnack House of the Max Planck Society. The host, Prof. Carmen Scheibenbogen, said she was happy with the strong media presence, including several German broadcasters and science writers.
The conference is also organized and funded by ME/CFS Research Foundation. Their chair, Jörg Heydecke, gave an introduction with an update of their economic report. It estimates that there are approximately 1.4 million patients with Long Covid and ME/CFS in Germany and that the yearly societal costs are as high as € 64 billion.
Genetic risk focused on the brain
The first presentation came from Prof. Chris Ponting, University of Edinburgh, and focused on the big genetic study called DecodeME. The main results have already been shared as a preprint, but haven’t formally been published in a journal yet. This is because the researchers want to conclude a couple of additional analyses first. These include the HLA region, which is important for autoimmunity, further ‘fine-mapping ‘to identify the causal genes in a genomic region, genetic correlations with other illnesses, comorbidity genetics, etc. These analyses are all nearly done except the one on the X chromosome. That one is still ongoing and might tell us why women are more likely to get ME/CFS than men.
The analyses seem to have strengthened Ponting’s confidence in the results. He now made a powerful statement that ME/CFS heritability is exclusively enriched in neural tissues. Ponting showed that the DNA analysis of ME/CFS has substantial overlap with fibromyalgia and that for both, the genetic risk is focused on the brain, and neurons in particular.
There was no direct genetic evidence for immune-related pathogenesis. They hoped to have found a genetic association in the HLA region because that would have been a strong signal that ME/CFS involves autoimmunity, but no such evidence was found.
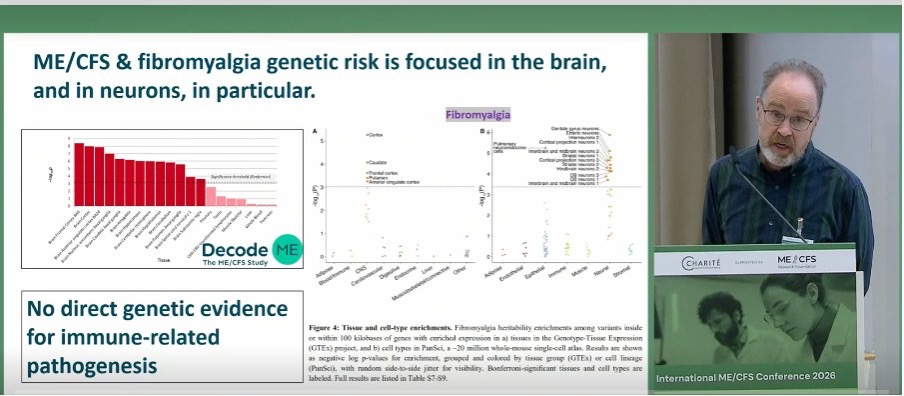
Someone asked if the genetics highlights certain cell-types in the nervous system: is it more microglia or more synaptic function? Ponting answered that they are doing this analysis right now, but “everything we have right now says it’s neurons, not glia. There are indications for synaptic protein genes being involved”, he said.
DecodeME also compared patients with high and low symptom burden based on 67 questionnaire responses, but did not find a significant difference in their genetics. Genes that came close to significance were RORA, SPP2/TRPM8, and RARRES2.
Lastly, Ponting pleaded for long-read sequencing of the HLA region to get a better resolution and an analysis of rare genetic variants. He hopes to get this done in the next big study called SequenceME. Interestingly, an analysis of rare variants from the UK Biobank already pointed to the gene BTN2A1, which is involved in T-cell activation. It also came up in DecodeME, so there could be an important lead for research on the immune system in ME/CFS after all.
If you would like to support this line of research, you can donate to SequenceME here:
https://www.actionforme.org.uk/research-campaigns/our-research-work/sequenceme-long-covid/donate-sequence-me/
Whole-body PET scans
Dr. Michelle James from Stanford University gave a fascinating talk on whole-body scans. Her work is based on positron emission tomography (PET). It involves injecting a radioactive molecule into the veins, called a tracer. Ideally, you want a tracer that is highly specific to a particular cell type or process so that you know exactly what the radioactive signal means. Dr. James works with a tracer for the translocator protein (TSPO), which binds to microglia, the brain’s immune cells.
PET scans with a TSPO signal have been used to study neuroinflammation in various disorders. But instead of looking only at the brain, James did PET scans of the entire body. The results showed striking differences between ME/CFS patients and controls. Most of the TSPO signal in ME/CFS patients was found in the shoulders, postural muscles, the neck, glutes, and also in the bone marrow. The data presented were from 15 female ME/CFS patients and matched controls. Interestingly, the researchers also had a pair of identical twins, one with and one without the disease.
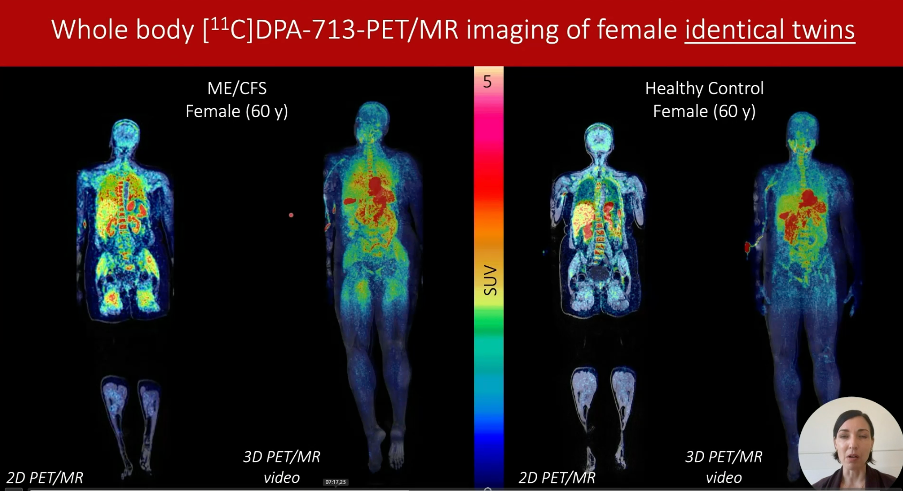
It’s not exactly clear what these images mean. TSPO is not just a marker of microglia activation, but also of mitochondrial function and cellular bioenergetics, so that might be what it’s picking up. Interestingly, when James looked at the brain, she found a decreased TSPO signal. This was a bit of a paradox. They expected an increased signal due to suspected microglia activation and low-grade inflammation in ME/CFS, but that wasn’t the case. How can the signal be increased in the muscles but not in the brain? James said something similar has been found before in schizophrenia and depression. She thinks it’s due to peripheral inflammation inhibiting the TSPO tracer from going to the brain. Her explanation was supported by plasma proteomics, which showed several inflammatory markers to be increased in her ME/CFS patients.
Diffusion MRI
Perhaps these PET results are related to another brain scan study presented by Qiang Yu from the University of the Sunshine Coast in Australia. His team used diffusion tensor imaging, which is based on how freely water molecules can move around and diffuse.
Yu’s results have already been published, but their interpretation has changed since then. In the paper, they thought they had found evidence of neuro-inflammation, but careful analysis (including by members of the Science for ME forum) seems to have changed their view.

During the presentation, Yu explained that the results show the ME/CFS brain is not experiencing edema of the blood vessels but rather a different form of swelling he called “cytotoxic-like” edema.
ME/CFS patients also had a reduced restricted fraction. This refers to water trapped inside cells and is a measure of immune cell infiltration. Because this restricted fraction was lower in ME/CFS, it did not suggest inflammatory lesions, but possibly shape changes or depletion of microglia. That might be consistent with a brain autopsy study from the Netherlands that also found signs of worn-out rather than overactive microglia.
Lastly, the Australian team also found an increased fibre fraction, which refers to water moving in a particular direction, such as along axons. According to Yu, increased fibre fraction may reflect increased density of the axons or a reduced extracellular space.
As you can hear, these diffusion metrics are all indirect measures, so there’s a lot of uncertainty in their interpretation. But things could get interesting if different imaging techniques, autopsies, and genetics start to align.
Immunoadsorption
The second day of the conference was focused on clinical interventions and treatment trials. Unfortunately, most did not bring good news. The most anticipated one tested immunoadsorption, a technique that filters antibodies from the blood.
Immunoadsorption has been reported to be helpful in ME/CFS patients before, but it hasn’t been tested in a randomized controlled trial yet. There are several such studies underway, including the IA-PACS-CFS trial conducted at the Charité in Berlin. Dr. Hannah Pressler and Dr. Friederike Ufer presented the first preliminary results. This study included a sham-control arm where patients received the immunoadsorption procedure, but without actually filtering out antibodies. In this group, the adsorbers were already saturated with antibodies so that they could no longer extract them from the blood. This study design with a control group gives us a more accurate way to test if the treatment really works.
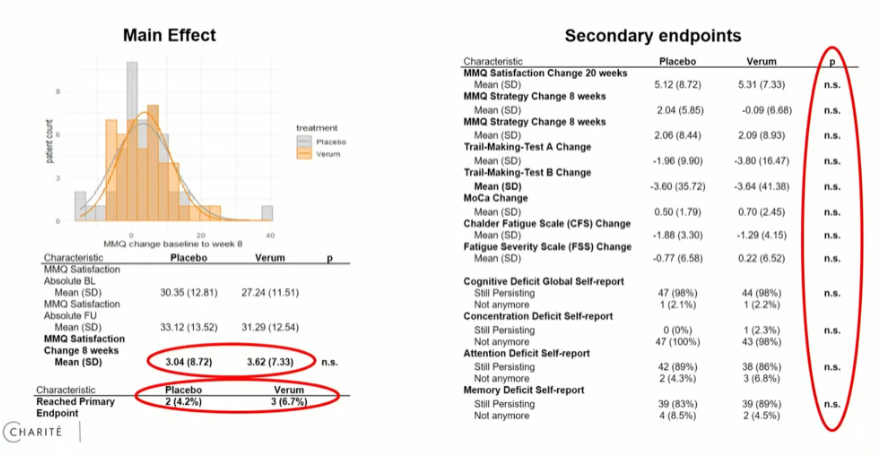
The results presented were from 45 patients in the treatment group and 20 in the sham-control group. All had ME/CFS triggered by COVID-19. On the primary outcome, the Chalder Fatigue Scale, the difference was -2.34 points (95% confidence interval -6.61 to 1.94 points). This means that the immunoadsorption group did a little bit better, but the difference was not significant (p-value: 0.278). The secondary outcome of physical function on the SF-36 questionnaire showed similar null results. The researchers concluded that the primary endpoint was not met, with no clear overall treatment effect.
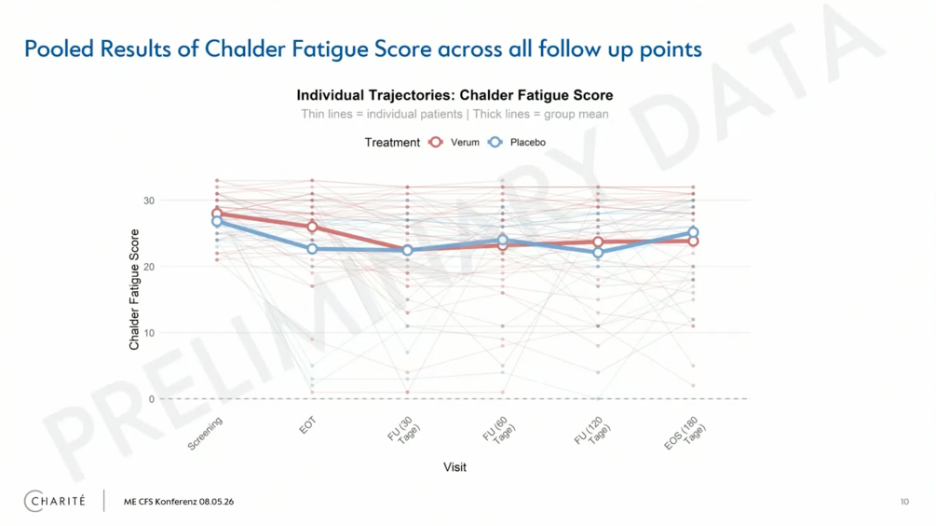
With the genetic analysis finding nothing in the HLA region, this was the second blow to the autoimmune hypothesis. But there are some caveats. Both Prof. Birgit Sawitzki and Prof. Carmen Scheibenbogen noted that ME/CFS is heterogeneous and that a subgroup might have had a positive response. The trial was underpowered to detect this because it had only 66 participants. The confidence interval for fatigue goes all the way up to a 6.6 point difference in favor of the immunoadsorption group. This means the treatment could have a modest effect, but the trial had too few participants to demonstrate this.
A stricter selection of participants based on immunological parameters (e.g., only those with high antibody titers) might be important for future studies. Prof. Sawitzki plans to do a detailed analysis of immunoadsorption responders to see if something sets them apart.
Daratumumab
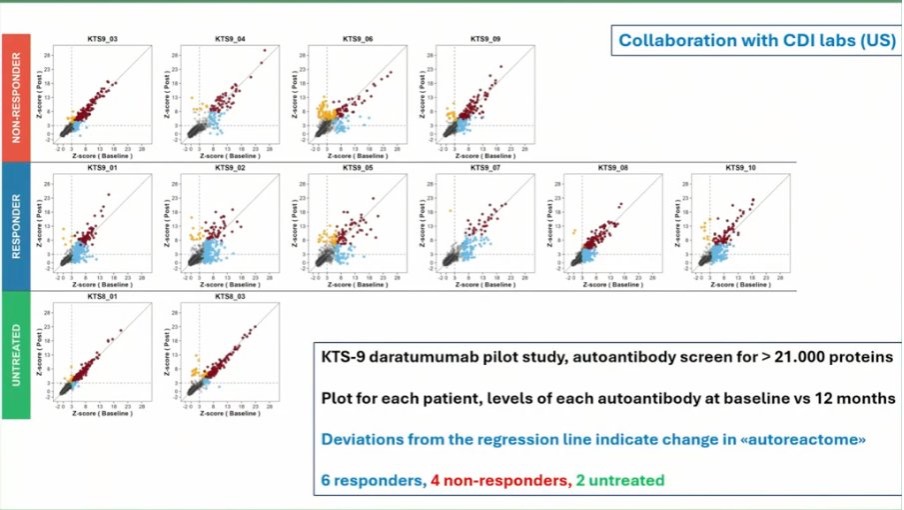
Another promising treatment in ME/CFS is the cancer drug daratumumab. It targets plasma cells that produce a lot of antibodies. At the Berlin conference, the Norwegian team of Øystein Fluge presented data on the pilot study of 10 ME/CFS patients on daratumumab. Fluge showed a graph where the 6 responders had a larger variation of their autoreactome (the collection of all their autoantibodies) than the others. The responders had more changes after treatment, suggesting a resetting might have taken place. The pilot study also showed that patients with more natural killer cells had a better response.
Fluge then gave a brief update on their randomized study of daratumumab called ResetME. They already included 38 out of their target of 66 patients, which is encouraging. Two-thirds will get the drug, the other third a placebo.
The Inclusion criteria are as follows:
- a diagnosis according to the Canadian Consensus Criteria (CCC)
- moderate to severe ME/CFS (mild is not included)
- more than 2 years of illness duration
- a baseline natural killer cell count higher than 125 (10^6/L)
17 patients have already completed the 8-week follow-up after treatment. There was one serious adverse event: a hospitalization for three days due to worsening of ME/CFS symptoms. We certainly look forward to hearing more about this study. Because of the promising results in the pilot study, it’s probably the most exciting drug trial in the ME/CFS field at the moment!
If you would like to support this daratumumab randomized trial, you can do so here:
https://www.me-foreningen.no/om-oss/stott-me-foreningen/me-fondet/english-me-fund/
Low-dose naltrexone
Dr. Luis Nacul presented the first results from his Canadian trial of low-dose naltrexone (LDN). It’s a drug to treat addiction to alcohol or opiates. In low doses, however, naltrexone is suspected to stimulate the body’s production of endorphins and influence the immune and central nervous system.
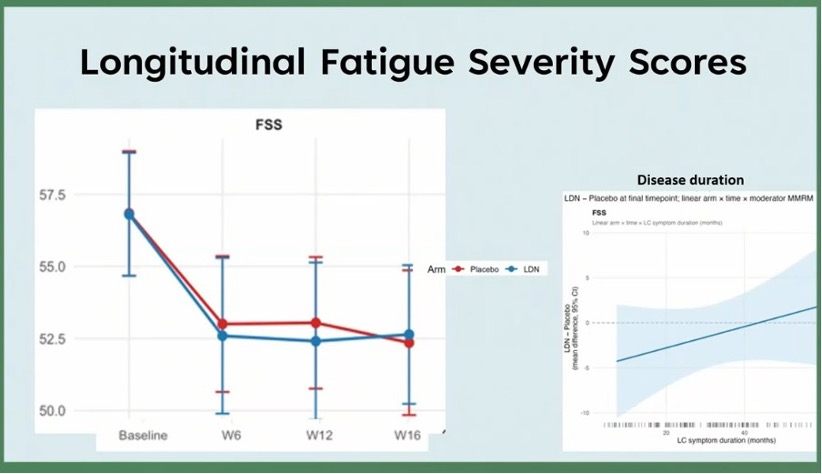
In Nacul’s study, the dose was titrated up from 1 to 4.5 mg of LDN per day. 160 patients were randomized to receive either LDN or a placebo. Unfortunately, at 16 weeks, fatigue scores showed no significant difference between the two groups. As in the immunoadsorption trial, it could be that certain subgroups respond better, but the primary outcome looks negative overall. [EDIT: other outcomes such as pain are expected to be analyzed and shared later]. Several other teams are also conducting trials of LDN in ME/CFS, so we will likely have more data on this medication soon.
Rehabilitation
Dr. Kristen Wittke presented the results of ‘CFS_CARE’, an interdisciplinary treatment for ME/CFS developed at the Charité Fatigue Center. It was created in collaboration with the German insurance funds with the aim of supporting and rehabilitating ME/CFS patients. Unfortunately, the results were rather negative. We weren’t able to watch this presentation in full, but the final slide summarized the findings forcefully, stating:
“An integrated multidisciplinary care model did not improve physical functioning even under tailored conditions. The linkage of participation in rehabilitation and social benefits must be reconsidered in patients with ME/CFS.”
Methylprednisolone
Lastly, Dr. Lucas Adam from the Charité shared (rather disappointing) results of PoCoVIT, a trial of methylprednisolone in patients with Long Covid. Methylprednisolone suppresses the immune system and is sometimes used in autoimmune diseases such as multiple sclerosis or lupus. Unfortunately, Dr. Adam had to stop his trial early due to many adverse effects, including 5 serious ones. At that point, only 96 Long Covid patients had been enrolled, while the recruitment goal was 418. Analysis of the results showed no significant difference for methylprednisolone compared to placebo on all outcomes.
There were many more interesting talks and discussions; this was merely a selection that caught our eye. An overview of all the speakers, abstracts, and presentations is available here. If there’s anything important that we’ve missed, feel free to post it in the comments section below.
You can support the work of the ME/CFS Research Foundation here:
https://mecfs-research.org/en/support/spendenabo/
Great work! I’d maybe add that we got the news that a second CD38 Mab (same as Daratumumab) will enter into a phase 2b trial this autumn and it will sponsored by sanofi. There will also be biomarker tracking and MRI imaging alongside it to identify and define responders.
Thanks for sharing this info! I should probably also add that Dr. Judith Bellmann explained that she will do a small (n = 38) randomized controlled trial of the anti-CD19 drug Inebilizumab in ME/CFS.
Thank you so much for your important reporting!
I’m sceptical of this sentence in the LDN section though. “ As in the immunoadsorption trial, it could be that certain subgroups respond better, but the primary outcome looks negative overall.”. Given the negative outcome for there to be a statistically significant subgroup of responders that might require torturing the data to the point there is a statistically significant subgroup of decliners. Seems to me more likely that in this cohort in the way it was administered LDN simply did not work.
As a patient with lupus, along with other MRI-confirmed preconditions and multiple genetic markers confirmed through 23andMe, I’m very pleased to see that this conference has made such significant progress.
Bei der LDN Studie wurde die Auswertung nur bzgl Fatique gemacht. Es sollen weitere Auswertungen zu anderen Endpunkten folgen, wie z.B. Muskelschmerzen.
Thanks. I’ve added this to the text.
Low-dose naltrexone (LDN) may help alleviate generalized muscle pain; however, it does not address the underlying etiology. The differential diagnosis remains broad and may include genetic factors such as ACTN3 variants, spinal stenosis with compromised cerebrospinal fluid (CSF) flow, Tarlov or other meningeal cysts, or potential sarcolemmal pathology.